Abstract
Introduction: High-dose Ara-C (HiDAC) is a post-remission consolidation protocol commonly used in patients with acute myeloid leukemia (AML). The optimal regimen of HiDAC administration is not fully established. Recent studies suggest that a short and condensed schedule of HiDAC consolidation therapy given on days 1, 2 and 3 (HiDAC123) may be superior to the traditional days 1, 3 and 5 regimen (HiDAC135). The current retrospective study compared the impact of these two therapeutic approaches on the outcomes of AML patients treated at the Rambam Leukemia Unit.
Methods: This retrospective cohort analysis included patients diagnosed with AML between the years 2015-2020 who were candidates for aggressive chemotherapy. Fifty-seven patients received a total of 76 courses of HiDAC135 mainly between 2015 and 2017, while 77 other patients received a total of 135 courses of HiDAC123 mainly between 2017 and 2020. The HiDAC dose in the two groups was 3 g/m 2 for patients aged less than 55 years, and 1.5 g/m 2 for patients aged 55-65 years. Patient demographics, the ELN leukemia risk category and NPM1 status were compared. The analysis also included data on complications per course of HiDAC therapy, need for hospitalization, the number of days of hospitalization if needed, the occurrence of neutropenic fever per course, the need for blood products (packed red blood cells and platelets), neurotoxicity and death within the 28 days of the course.
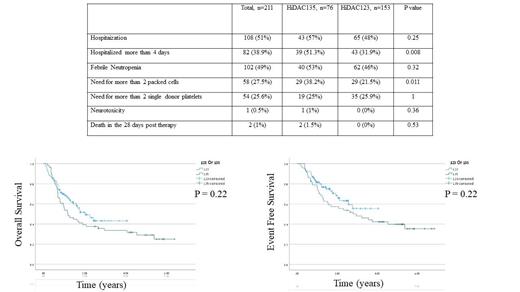
Results: The HiDAC123 and HiDAC135 groups were comparable in terms of the ELN risk category and NPM1 status. The groups significantly differed in age (median of 49.8 years and 55.9 years, respectively; p=0.01), which was most likely to be related to the use of venetoclax and azacitidine during the period when HiDAC123 was prescribed. Similar percentage of patients achieved complete remission (CR) after induction chemotherapy (93.5% and 94.7%, in the HiDAC123 and HiDAC135 groups, respectively). There were no differences in the overall survival (OS) and event-free survival (EFS) between the two groups (Figure 1). Significantly more patients in the HiDAC135 group were hospitalized for more than 4 days compared with the HiDAC123 group (51.3% vs 31.9%; p=0.008) and significantly more patients in the HiDAC135 group needed more than 2 units of packed red blood cells than in the HiDAC123 group (38.2% vs 21.5%, p=0.011). There was no difference in the percentage of patients in need of platelet transfusion. Likewise, there was no difference between the two groups in either the number of hospitalizations post-HiDAC therapy, the occurrence of neutropenic fever events or the incidence of death within the first 28 days after the HiDAC therapy.
Conclusions: The present analysis demonstrates that HiDAC123 is as safe and efficacious as HiDAC135 in terms of OS and EFS and is associated with shorter hospitalization and lower requirement for blood products. These results show that HiDAC123 could be the regimen of choice as consolidation therapy in AML patients.
Zuckerman: Orgenesis Inc.: Honoraria; BioSight Ltd: Honoraria; AbbVie: Honoraria; Janssen: Honoraria; Novartis: Honoraria; Gilead Sciences: Honoraria, Speakers Bureau; Cellect Biotechnology: Honoraria. Ofran: Pfizer: Consultancy; AbbVie: Consultancy; Medison Israel: Consultancy; Astellas: Consultancy; Janssen: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal